- IMAGEJ QUANTIFY HE STAINING MANUAL
- IMAGEJ QUANTIFY HE STAINING SOFTWARE
- IMAGEJ QUANTIFY HE STAINING SERIES
We tested the functionality of our method in deltoid muscle biopsies from a heterogeneous population of patients with histologically normal muscle (male, female, old, young, lean, obese), patients with pathological muscle, and a small set of pectoral samples. Our macro was designed to assess fiber morphometry in digital images of the entire muscle.
Here, we provide a reproducible and accurate automatic image analysis macro script that runs in an open-access platform (Fiji-ImageJ) and allows for the fast quantification and differentiation of human muscle fiber composition and histomorphometry in different physiological and pathological conditions.
IMAGEJ QUANTIFY HE STAINING SOFTWARE
An increasing amount of software for the automated analysis of muscle images has been recently developed, yet some of these require extensive human intervention, are not publicly available, or have been only tested in rodents or are not designed for the analysis of human muscle in medical settings. However, this procedure is time-consuming, labor intensive, and subjected to user bias and may not adequately represent the whole tissue complexity and/or heterogeneity.
IMAGEJ QUANTIFY HE STAINING MANUAL
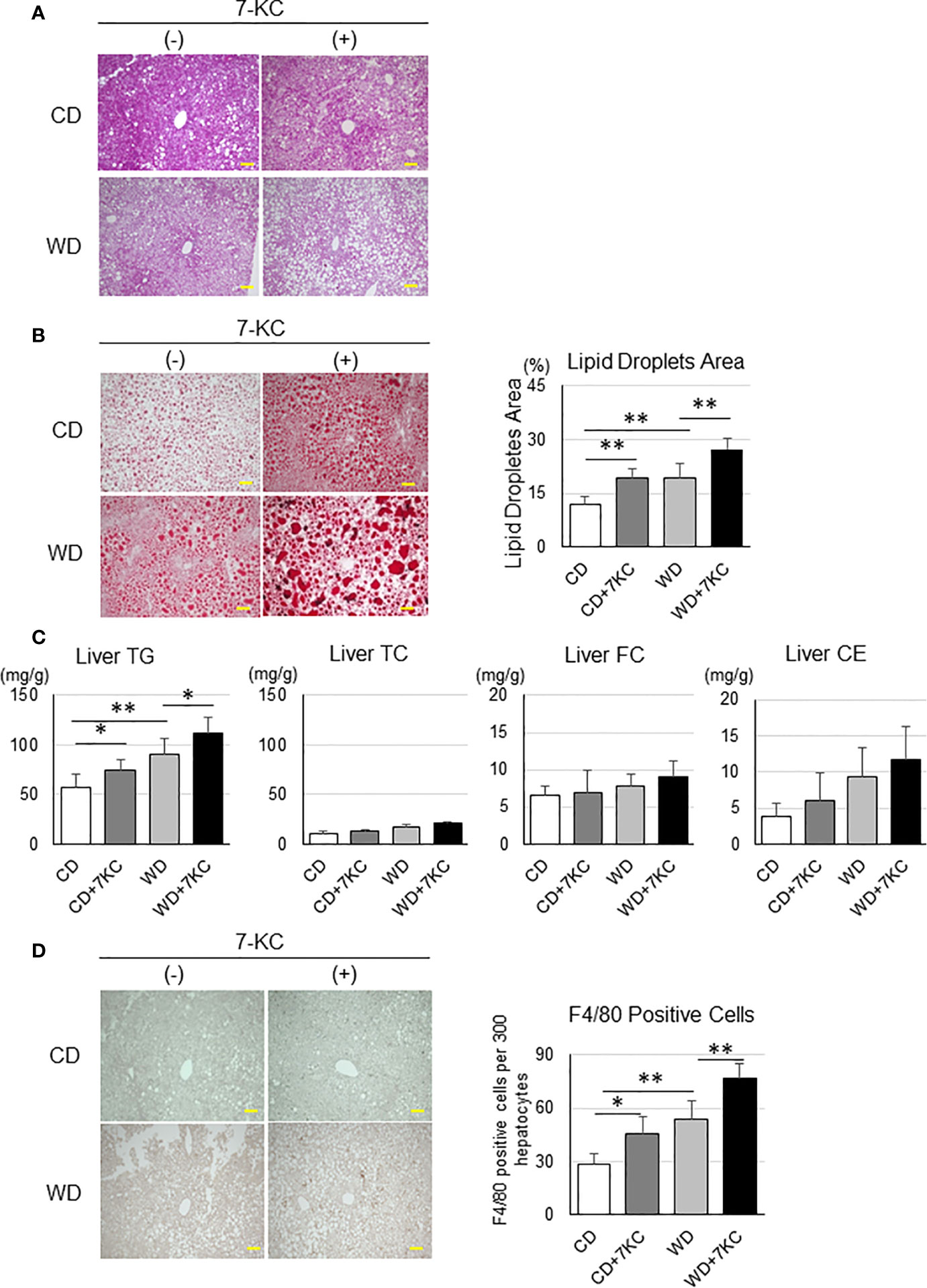
Because of the large number of myofibers within a muscle section, morphology evaluation by manual methods is usually limited to a small representative area (~ 400 fibers). The conventional procedure for quantifying muscle atrophy in fixed or frozen tissue consists of manual quantification of the myofibers’ size (fiber diameter at its narrowest point) to evaluate muscle morphology. Physiological aging and pathological muscle atrophies are characterized by a reduction of myofibers’ size and number, increased myofibers’ size variation, and alterations in type I (slow twitch)/type II (fast twitch) myofiber ratio. In the last few years, the growing development of innovative treatments, including biotherapies, requires a more stringent stratification of patients, with accurate quantification of myopathological processes. For decades, myopathological diagnosis remained based on purely qualitative evaluations, notably depending on pathologists’ experience. Muscle histology analyses are very effective for the characterization of morphological parameters to determine muscular health status and an important tool for the diagnosis of numerous muscle diseases. Our macro is reliable and suitable for the study of human skeletal muscle for research and for diagnosis in clinical settings providing reproducible and consistent analysis when the time is of the utmost importance. The output of the analyses includes excel files with the quantification of fibers’ morphometry and color-coded maps based on the fiber’s size, which proved to be an advantageous feature for the fast and easy visual identification of location-specific atrophy and a potential tool for medical diagnosis. The performance of the macro analysis was maintained in pectoral and deltoid samples from subjects of different age, gender, body weight, and muscle status. Likewise, our tool showed high accuracy, as compared with manual methods, for identifying the total number of fibers ( r = 0.97, p < 0.001), fiber I and fiber II proportion ( r = 0.92, p < 0.001), and minor diameter ( r = 0.86, p < 0.001) while conducting analysis in ~ 5 min/sample.
IMAGEJ QUANTIFY HE STAINING SERIES
Our macro is fully automated, requires no user intervention, and demonstrated improved fiber segmentation by running a series of image pre-processing steps before the analysis. We tested the functionality of our method in deltoid muscle biopsies from a heterogeneous population of subjects with histologically normal muscle (male, female, old, young, lean, obese) and patients with dermatomyositis, necrotizing autoimmune myopathy, and anti-synthetase syndrome myopathy. We developed a new macro script for Fiji-ImageJ to automatically assess human fiber morphometry in digital images of the entire muscle. Manual and semi-automated methods to assess muscle morphometry in sections are time-consuming, limited to a small field of analysis, and susceptible to bias, while most automated methods have been only tested in rodent muscle. Accurate and stringent assessment of myofibers’ changes in size and number, and alterations in the proportion of oxidative (type I) and glycolytic (type II) fibers is essential for the appropriate study of aging and pathological muscle, as well as for diagnosis and follow-up of muscle diseases. The quantitative analysis of muscle histomorphometry has been growing in importance in both research and clinical settings.
